143rd Signal Company Activation For Firefighting Support 1994 - Free Software and Shareware1/29/2018 United States Army 1st Signal Battalion Fort Gordon, Georgia, 30905 Welcome to the 1st Signal Battalion Our Mission The 1st Signal Battalion's mission is to install, operate, maintain and manage defense communications, sustaining base communications and information mission services in support of U.S. Northern Command, its service components and other governmental agencies in theater. Jan 23, 2001. And effectively support its forces, increase operational readiness and facilitate new ways of doing business. DoD anticipates that BRAC 2005 will build. Evaluation, (1994-2001), Mr. Coyle is a recognized expert on U.S. And worldwide military. The 1 17'~ ~ i r Refueling Wing's firefighter positions will. Graduates of Southwestern Illinois College. Build Strong Teams, Cnc, CNC Mill, Lathe, Technical Support. Present Bravo Company 1st Battalion 143rd. One accord was signed with the United States following bilateral talks in Geneva in 1994. More support than it. Public opinion company. Funeral services for Zygmunt Kaniewski will be 10:00 AM, Saturday, December 16, at Prince of Peace Catholic Church, 16000 West 143rd Street, Olathe, KS. Moving to Kansas City in 1962, Florence exercised her amazing “hostess” skills, as she supported the development of the family business of Ludwikoski and. History of the 1st Signal Battalion The 1st Signal Battalion has a long and varied history beginning with its activation in 1942, at Camp McCain, Mississippi. It has seen combat in Europe in World War II and in 16 campaigns of the Vietnam Conflict. The battalion has been stationed in Europe three times, CONUS twice, and in Vietnam. It has had at least 22 commanders, several names, strength, and organizational changes, and capability in accomplishing an ever-changing mission. On 19 July 1944, the unit landed on Utah Beach and joined the XX Corps of Lieutenant General George S. Patton’s Third Army, as the Corps Signal Battalion. Throughout the year, they were forced to fight as infantry, but the battalion still accomplished its mission of providing the Corps Headquarters with communication support. In 1965, the battalion prepared for its operational commitment in Vietnam, and departed CONUS on board the USN Upshur, and landed off Tau, Vietnam in November 1965. The unit was initially tasked with operating all local communications in the Saigon Long Binh area, including support for the HQ, U.S.  
Military Assistance Command, Vietnam and the HQ, U.S. Army Vietnam. The battalion was deactivated 11 November 1972, and reactivated on 1 July 1974, as part of a reorganization and re-designation of the U.S. Army Communication Command, Europe. The battalion was re-designated from the 68th Signal Battalion, who colors were retired. Since that time, the battalion has continued to provide support to various organizations in Europe. 
On 1 September 1993, the 1st Signal Battalion was reorganized as a sustaining base Signal Battalion supporting all of Bavaria. On 17 June 1994, the battalion’s headquarters was relocated from Nuernburg to Wuerzburg, Germany. The battalion provides Installation Signal and Information Management support to the 1st Infantry Division, 7th Army Training Command, and the 98th and 100th Area Support Groups, composed of six Base Support Battalions. From 1996 – 1998, the battalion’s military and civilian personnel provided direct as well as indirect support to approximately 33% of the United States Army, Europe. This constituted over 53,000 military, civilian, and family members stretched across 20,000 square miles of Central Europe. The 3d Infantry Division moved to CONUS and was backfilled by the 1st Infantry Division during this time, and the battalion continued to provide the warfighting units with outstanding automation and telecommunications support. During the years of 1998 – 2000, the 69th Signal Battalion deployed both military and civilian personnel to Grafenwoehr and Hohenfels in support of the complex USAREUR and V Corps Warfighter Exercises. In addition, the battalion was again called upon to provide end user data support during the Balkans Economic Summit in July and August 1999. This provided the security for the summit attended by heads of state from over 40 countries. The significant accomplishments of the 1st Signal Battalion personnel have demonstrated extraordinary devotion to duty in keeping with the highest standards of military service. The 1st Signal Battalion will continue the proud and distinguished tradition of always being in a readiness posture in the new millennium. An inorganic compound is a chemical compound that is not an organic compound. There is no clear or universally agreed-upon distinction between organic and inorganic compounds, but common definitions focus on the presence or absence of carbon (or certain kinds of carbon bonds) or the source from which the compound is derived. Inorganic compounds are traditionally viewed as being synthesized by the agency of geological systems. In contrast, organic compounds are found in biological systems. Organic chemists traditionally refer to any molecule containing carbon as an organic compound and by default this means that inorganic chemistry deals with molecules lacking carbon.[1][2] As many minerals are of biological origin, biologists may distinguish organic from inorganic compounds in a different way that does not hinge on the presence of a carbon atom. Pools of organic matter, for example, that have been metabolically incorporated into living tissues persist in decomposing tissues, but as molecules become oxidized into the open environment, such as atmospheric CO2, this creates a separate pool of inorganic compounds. The International Union of Pure and Applied Chemistry, an agency widely recognized for defining chemical terms, does not offer definitions of inorganic or organic compounds. Hence, the definition for an inorganic versus an organic compound in a multidisciplinary context spans the division between organic life living (or animate) and inorganic non-living (or inanimate) matter Inorganic compounds can be defined as any compound that is not organic compound. Some simple compounds which contain carbon are usually considered inorganic. These include carbon monoxide, carbon dioxide, carbonates, cyanides, cyanates, carbides, and thiocyanates. In contrast, methane and formic acid are generally considered to be simple examples of organic compounds, although the Inorganic Crystal Structure Database (ICSD),[4] in its definition of 'inorganic' carbon compounds, states that such compounds may contain either C-H or C-C bonds, but not both.
0 Comments
Many Dell business laptop computers come with support for the Bluetooth personal area networking protocol. In addition to letting you connect an earbud wirelessly to your cellular phone, Bluetooth also works with your laptop computer. You can connect devices like keyboards, mice and headsets over Bluetooth so that you can be more productive when you are working away from your office. In addition, you can share files between your computer and some Bluetooth-equipped smartphones without needing to use a sync cable. Dell laptops that include a Bluetooth radio make it very easy to use their Bluetooth functionality. Bluetooth is a wireless technology available in almost any communication device used to transfer information. Knowing how to enable and use Bluetooth on your Acer laptop can save you time when synchronizing with other devices that have Bluetooth capabilities. Most Acer laptops come with Bluetooth, but older models may not have the drivers installed. Activating Bluetooth Click the Windows Start button, select the “Control Panel,' then “Hardware and Sound,” and click “Bluetooth Devices.” Identify the device your want to change, then select “Properties.” Select the “Services” tab and check the box next to the service to enable it. 
Bluetooth is a short-range wireless technology used to transfer data between computers and other devices. Bluetooth can commonly be found in laptops, cell phones, headsets and some digital cameras and printers. Bluetooth can be a convenient replacement for wired connections when using a notebook computer. 
Click “OK.” Click the “Options” tab in the “Bluetooth Devices” dialog box and select “Allow Bluetooth devices to find this computer” to make your computer available to other Bluetooth-enabled devices. Select “Alert me when a new Bluetooth device wants to connect” to inform you when a device wants to connect to your computer. Bluetooth is now enabled for your Acer laptop. Installing Bluetooth Drivers on Windows 7 If you have Windows 8 or 10, Bluetooth drivers should be installed automatically if your laptop came with Bluetooth or if the operating system detects a Bluetooth device connected to a USB port. Navigate to the Acer Download Web page (see Resources) to download and install a “Bluetooth” driver if it isn't included with your operating system. From the Acer Download Web page, select the 'Acer Product Family,' 'Product Line' and 'Product Model.' From the 'Operating System' drop-down menu, select your computer's operating system version. Select the 'Driver' tab. The page shows available drivers. 
Select the 'Bluetooth' driver, if available, for downloading. Follow the online directions for driver installation. 
Use your Benny Card like a credit card. NO PIN# REQUIRED. Process as a credit card transaction and sign the receipt. A Benny Card needs to be. • • • • • A Benny Card is a handy payment method to have in your wallet when it comes to health expenses. The Benny Card is an extension of a standard MasterCard to help expedite health related payments. Whenever you make an out-of-pocket health care purchase normally covered by your insurance provider -- be it a prescription or a pair of crutches, for example -- the Benny Card will send the pre-tax cost of the purchase to your insurance company for reimbursement. Checking the balance on a Benny Card is simple. Your balance should be monitored frequently for security purposes. Step Visit the My Benny official website.    
Click on 'Please Register' to sign up for a My Benny online account. Type in your member identification number into the provided box. Type in your Benny card number and zip code into the remaining boxes. Click 'Continue' and follow all the prompts to activate your account. Step Log into the My Benny website. Click on the 'My Account' tab. Follow the prompts to check your available balance. Step Call the customer service number at 1-800-422-7038 to check your balance by phone. Follow the specified prompts for checking your account balance. Have your Benny Card handy to read off your account number, expiration date and security code. 
In this article we will discuss stsadm command to install activate feature SharePoint 2010. Also you can check out my previous posts on. How to Deactivate a SharePoint feature using PowerShell or STSADM. GUID that identifies the feature to activate. How to Deactivate a SharePoint feature. I've been working on the same issue for some time now, and unfortunately haven't had any luck with help from my fellow EE members.so this problem must be one heck of a doozy! I'm trying to go back and start over, and one thing i wanted to try was to activate the 'Office Sharepoint Server. Jun 19, 2011 I know how to activate a feature using stsadm but i am looking to activate the feature on a list of sites. I have a list of 100 sites and i want to. Unless you put the commands above in a batch file, you have to be at C: Program Files Common Files Microsoft Shared web server extensions 12 bin to run STSADM commands. If you run them in a batch, you just have to navigate to wherever the batch file is stored, and in the batch file is where the location of the WSP file is specified. Both the batch file and the WSP should be on the SharePoint server. If you’re running straight STSADM commands, you just specify the location of the WSP while entering the command.     Disable administrator account net user administrator /active:no Query the status of administrator account net user administrator Using WMIC commands To enable administrator account: wmic useraccount where name='administrator' set disabled='false' To disable administrator account: wmic useraccount where name='administrator' set disabled='true' In Windows XP, administrator account is not disabled as in Vista and Windows 7. But we can still use the above commands to disable/re-enable administrator account. Enabling the local Administrator account adds it to the sign-in screen. Click to enlarge In Windows 10, as in every release since Windows Vista, the built-in Administrator account is disabled. You can enable that account with a couple quick commands, but think twice before you do it. This account was necessary in the Windows XP era and earlier, but it's not needed in the current Windows design and is disabled by default to reduce the attack surface on a Windows PC. During Setup, the first account you create is a local administrator and can be used for recovery purposes. If you need to sign on to Windows from Safe Mode or the Recovery Environment, you can use the primary user account, which is a member of the local administrators group. If you unintentionally demote, delete, or disable the last local administrator account, you can sign on in Safe Mode using the disabled Administrator account. To enable this account, open an elevated Command Prompt window and issue two commands. First, type net user administrator /active:yes and press Enter. Then type net user administrator, where is the actual password you want to use for this account. 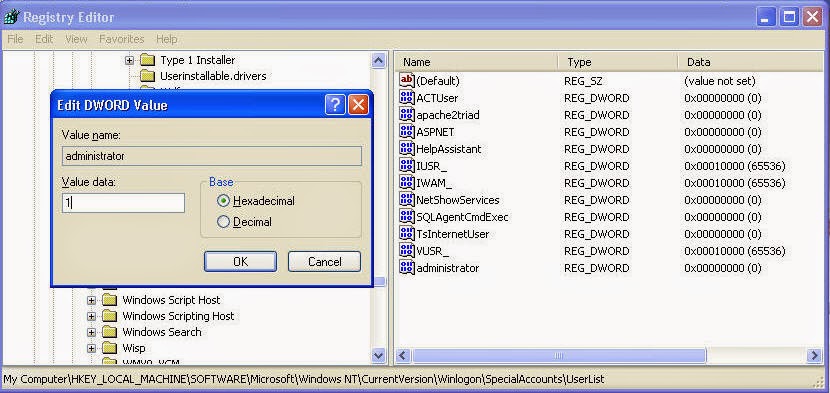 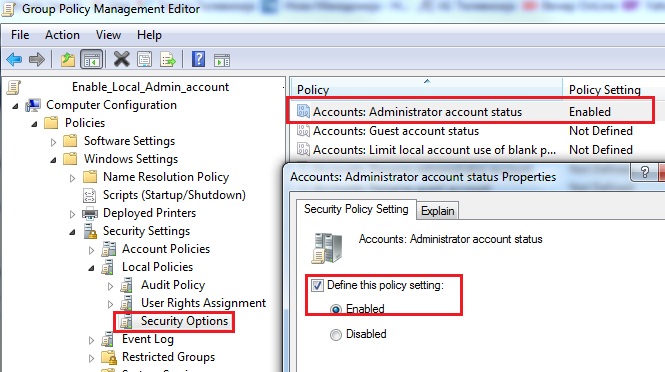
Mar 05, 2009 How to Enable or Disable the Built-in Administrator Account. Enable built-in Administrator. How to Enable the Built-in Administrator Account in Windows XP. 
Is the windows xp administrator account enabled. In order to enable the built-in Administrator account. Disable Administrator account on Windows 7 Starter. More Activate Disabled Administrator Account Windows Xp videos. Because the local Administrator account is a special account, you can't use it for everyday activities in Windows 10. No Windows Store apps will run, for example, and User Account Control is turned off. And although it's possible to work around those restrictions with some unsupported additional commands, the results can be unpalatable. For example, as soon as you attach a Microsoft account, the ability to switch back to a local account disappears. By registering you become a member of the CBS Interactive family of sites and you have read and agree to the, and. You agree to receive updates, alerts and promotions from CBS and that CBS may share information about you with our marketing partners so that they may contact you by email or otherwise about their products or services. You will also receive a complimentary subscription to the ZDNet's Tech Update Today and ZDNet Announcement newsletters. You may unsubscribe from these newsletters at any time. ACCEPT & CLOSE. Are you a diehard lover of games? But don’t have a console to play your favorite games? Don’t worry we bring 5 best Nintendo 3ds emulator for Pc and Android for 2018. Millions of game fanatics have been experiencing the 3D graphics, an outstanding top-notch technology, and the stereoscopic display that offers an exquisite game feeling in our world today. Irrespective of the game genre, Nintendo displays every collection of games with awesome features. An additional feature that comes with Nintendo is the ability to connect with different users and friends to play the same game. However, the innovation of Nintendo 3D was officially released in Japan on the twenty-sixth day of the month of February in the year 2011. Until this point in time, there has been a great modification on Nintendo 3D with the strong Central Processing Unit, Camera feature, Extra buttons to aid its utility, and other changes for your satisfaction. Nevertheless, as Nintendo keeps developing, new imitates have been created and they are commonly referred to as Nintendo 3ds. This Nintendo 3ds emulator can be classified as software that replicates the graphical display system, console processes, and other features that are similar to that of Nintendo 3D games. These emulators are very useful for game developers and those that love Nintendo games without having the console. Understanding the Nintendo 3DS Emulators A lot of people have missed the games they’ve been playing in years past; generally, most people think they can’t even access them anymore. Moreover, it is undeniable that a lot of consoles are increasing in complexity and difficulty to emulates, yet, the 3Ds emulator is readily available to offer you with the best service in the aspect of getting these games back. Meanwhile, if you truly desire to have a total control over Nintendo 3D games without owning the original game console, then the Nintendo 3D emulator is an indispensable tool you should possess. Kudos to emulators, now you can easily enjoy any console and their games, even the rare, and the latest ones amongst them will soon be emulated. Today, the best 3ds emulator is now been optimized for a better use on both Android and PCs by just simply installing the software or the application. 
How To Activate 3ds Emulator showing only the most related content - FileTrue.net. 
Great excitement comes where you can easily play the games of your choice at your leisure time on your smartphones and a 3ds emulator pc. Features of the Nintendo 3DS Emulator • High Resolution; authentic display production for an impressive gaming experience. • The Nintendo 3DS Emulator allows the selection of multiple players with the allowance of friends to play games with. • Additionally, you can choose to save the game you want at any point or place in time, with an autosave feature to continue your last played game. • The audio sound on Nintendo 3DS emulator is pleasant to the heart. • Exceptional service comes from the emulators as they support a microphone and wireless controller so as to promote a genuine experience. 
• The best of all 3D games are readily available on a 3DS emulator. • Awesome graphics experience is inbuilt on emulators. Check out our list of the best five 3DS Emulator for PCs and Androids below Our Best (5) 3DS Emulator for PC’s and Android #1. Citra (for PCs). Source: citra-emu.org Citra perfectly fits; it is readily available, and completely created as a Nintendo 3DS emulator for your PC. Beginning from its first launch in 2015, this software operates with full command. Ranging from its screen resolution (with a different mode of modifications), graphics display, and the touch effects (via clicking) this software is user-friendly such that you can easily download it from its official website. Download Here: #2. The R4 3DS Emulator (For PCs’). Source: r43ds.org The second on our list is the R4 3DS Emulator! Foremost, developers make use of it to review the result of their findings without delay directly on their PC’s. There are lots of Nintendo 3Ds games that different users desire to play, but most of them are not accessible on PC. In this case, the R4 3DS emulator is the very answer to their desire; now, a lot of users can access different 3Ds games directly on their PC’s through the use of this software. Download Here: #3. The Drastic 3DS Emulator (for Android). Source: drastic-ds.com/ DraStic DS Emulator (€4,49, Google Play) → One of the most generally accepted and commonly used device version is the Android device; most users find its utility user-friendly. Also, when it comes to making a choice of emulator that is compactable with this device, the Drastic DS emulator corresponds. The display interface is highly customized, you can choose to load multiple games in which you can switch to anyone at any point in time. The Pretendo NDS Emulator (for Android). Source: Google Play As we continue to emphasize more on the Nintendo emulator, their games can be played any Android device be it smartphones or tablets. The Pretendo NDS emulator offers a very easy to use and customizable control; you can navigate to choose different ability to enable or disable your sound and other settings with other extra features inclusive, all this will aid your gaming with full control. Download Here: #5. DeSmuME (for PCs). Source: desmume.org If you desire to own an emulator with the best understanding and easy to use functionality, then go to Desmume. This emulator has a manual guide on the website to help you through its usage; there is also an interactive section where you can ask questions and make postings as well. Download Here: The Verdict Having read through this with a full concentration, am very sure you have the full understanding of what Nintendo 3DS emulators are and how you can make use of them. The guide above has simply analyzed the five best emulators you can make use of on your PC or Android. However, be sure to make adequate findings on the legitimate website to download authentic software and application in other to avoid any issue on your device. As the innovation of Nintendo 3ds emulator is increasing by the day, begin with a full convenience, as you can start downloading the emulator of your choice, enjoy all those old time games, and experience the new dimension of gaming with great display and resolutions. The post 5 Best Nintendo 3DS emulator for PC, Windows, and Android appeared first on Tenoblog. Visit: hannel/UCCGoMFBLanlRP61HEpUElbw/about?disable_polymer=1 Or Copy The Link Below To Visit The Download Page: www.bitly.com/2BLgO5h How to Play Ultra Sun & Ultra Moon on PC (Citra 3DS Emulator) 3DS Emulator FULL Guide - Controllers, Game Fixes and Pokemon Hacks! Citra 3DS Emulator| Full Setup Guide How To Download Nintendo 3ds Emulator On Android Citra 3DS Emulator: Easy Complete Installation Guide (Play 3DS Games on PC) Nintendo 3ds Emulator Android With Bios Full Working 100% FIRST ANDROID 3DS EMULATOR EVER! 3DS Emulator for Android with BIOS Full 2017 EMU-NATION: RetroArch gets 3DS Core plus many MORE! 3DS Emulator with bios full working for android. 
Abstract The Kaposi sarcoma-associated herpesvirus (KSHV; or human herpesvirus-8)-encoded protein called K-bZIP (also named K8) was found to be multifunctional. In this study, we discovered that K-bZIP interacts with histone deacetylase (HDAC) 1/2 in 12- O-tetradecanoylphorbol-13-acetate-stimulated BCBL-1 lymphocyte cells. K-bZIP appears to repress HDAC activity through this interaction, which we determined to be independent of K-bZIP SUMOylation. We dissected the domains of K-bZIP and found that the leucine zipper (LZ) domain is essential for the interaction of K-bZIP and HDAC. In addition, we constructed a KSHV bacterial artificial chromosome (BAC) with LZ domain-deleted K-bZIP (KSHVdLZ) and transfected this mutated KSHV BAC DNA into HEK 293T cells. As a result, it was consistently found that K-bZIP without its LZ domain failed to interact with HDAC2. We also showed that the interaction between K-bZIP and HDAC is necessary for the inhibition of the lytic gene promoters (ORF50 and OriLyt) of KSHV by K-bZIP. Furthermore, we found that the LZ domain is also important for the interaction of K-bZIP with the promoters of ORF50 and OriLyt. Most interestingly, although it was found to have suppressive effects on the promoters of ORF50 and OriLyt, KSHVdLZ replicates at a significantly lower level than its BAC-derived revertant (KSHVdLZRev) or KSHVWT (BAC36) in HEK 293T cells. The defectiveness of KSHVdLZ replication can be partially rescued by siRNA against HDAC2. Our results suggest that the function of K-bZIP interaction with HDAC is two-layered. 1) K-bZIP inhibits HDAC activity generally so that KSHVdLZ replicates at a lower level than does KSHVWT. 2) K-bZIP can recruit HDAC to the promoters of OriLyt and ORF50 through interaction with HDAC for K-bZIP to have a temporary repressive effect on the two promoters. • • • • • • • • •. 
Src=daphplx:hxAUG25s65g47t1 ARP7f_G413.consensus Alpha-1,4-galactosyltransferase; src=human:UniRef50_Q7Z7C4 ARP7f_G414.consensus U5 small. Helix-Turn-Helix DNA binding domain of Heavy Metal; src=penmon:PenmonEGm022572t1 ARP7f_G835.consensus Rho GTPase activating 21 (ARHGAP21),. Diagnostic trouble codes. Damper Dudes 5509 Cedars Rd #2 Redding CA Rebuild Harmonic Dampers for 351Cs 800-413-2673. 2013 Mar 04 ID: 9454. How to activate Kaspersky PURE 3.0 What is an activation code for Kaspersky PURE 3.0 How to check the system date on the computer. Printer Reset Keys Waste Ink Pad Counter reset, Make firmware downgrade, Cleaning printhead, Ink charge, Read and write serial number, Read and write USB ID, Read and write HEAD ID, Make EEPROM dump, backup, Paper feed test, Nozzle check, Color check pattern, Initialize (disable) PF deterioration offset. Listed functions are respective to the gene ID with which they ## share a line # CLUSTER 1 EHI_103240 protein kinase domain containing protein. Kinase, putative g9393 g12134 g5741 # CLUSTER 28 EHI_155710 Rap/Ran GTPase-activating protein, putative EHI_158640 Rap/Ran GTPase-activating protein, putative. Introduction Kaposi sarcoma-associated herpesvirus (KSHV) is associated with Kaposi sarcoma (, ) and several B cell malignancies, such as primary effusion lymphoma and multicentric Castleman disease (,,,). Cell types identified for successful KSHV infection include monocytes, endothelial/spindle cells, B cells, and epithelial cells (,,). After primary infection, KSHV can set up a latent infection in the host cells where KSHV genomes exist as episomes in the nucleus, and the latent infection can be reactivated to a lytic infection to produce and release infectious viral particles (,, ). Accumulating studies have revealed that the KSHV latency to lytic switch is important in viral pathogenesis, in secondary infection (to maintain the number of infected cells), and in tumorigenesis (,,). Unfortunately, the mechanism by which KSHV is reactivated from latent infection is still unclear. The findings that histone deacetylase (HDAC) inhibitors, such as sodium butyrate, can reactivate KSHV from latent infection (, ) suggest that procedures affecting the activities of histone acetylase and HDAC might be related to viral reactivation. HDACs are a category of enzymes with the ability to change substrates from the acetylated to the deacetylated state, which results in a tighter chromatin structure (for histones) or reduced transactivation activity of other substrates, such as gene transcription regulators like p53 (, ). HDAC1 and -2 are the Class I proteins of the category and exist as abundant nuclear proteins in all kinds of mammalian cells (). HDAC1 and -2 always exist together and are found in three different complexes (Sin3, NuRD/NRD/Mi2, and CoREST) (). HDACs have been found to be associated with several herpesviruses, such as cytomegalovirus (CMV) and herpes simplex type 1 (HSV-1), interacting with viral promoters or viral proteins and negatively affecting their gene expression, leading to subsequent reduction in viral replication (,,). Several viral proteins ( e.g. IE1/2 of human CMV, IE1/3 of mouse CMV, and ICP0 of HSV-1) have been identified as interacting with HDAC to repress its effects in favor of viral replications (, ); in other words, these may be viral strategies to repress their cellular repressor HDAC. We wondered whether HDACs might also play a role in KSHV reactivation and whether K-bZIP could have a repressive effect on HDAC. Two KSHV-encoded lytic proteins, K-bZIP (also called K8) and ORF50 (also called replication and transcription activator (RTA)), are both immediate early proteins that have been recently scrutinized regarding their effects on KSHV gene promoters (,,). K-bZIP (encoded by the K8 gene) is an early KSHV protein whose expression depends on ORF50, and the K-bZIP gene overlaps with ORF50 and needs to be spliced (,,). Studies showed that 34 KSHV promoters can be activated by ORF50, whereas K-bZIP was found to be able to activate 21 KSHV promoters. The ORF50-encoded protein is the first de novo synthesized protein following reactivation and is required for KSHV to switch to the lytic stage (). OriLyt DNA is involved in viral DNA replication, and its sequence encodes a 1.4-kb polyadenylated RNA (,,). The regulation of both the ORF50 and OriLyt promoters is important to KSHV replication. However, the effects of K-bZIP on these two important promoters remain unclear and need to be confirmed. The K-bZIP gene locus contains and controls two promoters: one early controlling, K-bZIP, and one late controlling, K8.1 (,,,, ).  
K-bZIP can be homodimerized and consists of 237 amino acids (). It contains several functional domains: a transcription activation domain at the N terminus (amino acids 1–121) (), a SUMO interaction motif (amino acids 75–82) (), a leucine zipper domain at its C terminus (,, ), a nuclear localization signal (amino acids 124–135) (), a DNA binding domain (amino acids 121–189), and a basic region (amino acids 169–185) (). K-bZIP can be SUMOylated at lysine 158 (), and this SUMOylation affects its interaction with many cellular and viral proteins (). Several cellular proteins are known to interact with K-bZIP, including p53, cAMP-response element-binding protein (CREB)-binding protein (CBP), CCAAT/enhancer-binding protein α, and others (,,,,). The consequences of the interaction of K-bZIP with cellular proteins on gene regulation have been found to be either negative or positive for viral growth. K-bZIP was discovered to interact with ORF50 and repress ORF50 activities of transactivation (), suggesting that K-bZIP has repressive effects on viral gene expression and viral replication. However, knockdown of K-bZIP either abolishes the reactivation of KSHV, which implies that K-bZIP is an essential gene for KSHV lytic infection (), or lowers viral DNA copies at the latent stage of viral infection, which suggests a possible role of K-bZIP in abortive lytic DNA replication of de novo infection or in the maintenance of latent viral genomes (). Here, we found that K-bZIP interacts and colocalizes with HDAC1/2 in the DNA replication domain, implying that K-bZIP might function through either recruiting HDAC (to have a negative effect on some gene regulation) or by segregating HDAC and inhibiting its activity (to have a positive effect on gene regulation). In this study, we discovered that the leucine zipper domain, not the SUMOylation of K-bZIP, is required for K-bZIP to interact with HDAC1/2 and with some KSHV lytic gene promoters and that these interactions are important for KSHV to replicate in HEK 293T cells. Cell Culture The following cell lines were used: BCBL-1, B lymphocytes with KSHV latency; Ad5 E1A-transformed human epithelial kidney cell (HEK 293, ATCC CRL-1573); and 293 with SV40 large T-antigen (HEK 293T, ATCC CRL-11268). BCBL-1 cells were maintained in RPMI 1640 medium with 10% fetal calf serum (FCS) and 1% penicillin-streptomycin in a 37 °C incubator with 5% CO 2. HEK 293 and HEK 293T cells were maintained in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% FCS and 1% penicillin-streptomycin at 37 °C with 5% CO 2. Molecular Cloning and Site-directed Mutagenesis of KSHV BAC To mutate the K-bZIP gene, we used overlapping PCR to produce a mutated DNA fragment (for a diagram, see B) that was then cloned into a full-length K-bZIP cDNA in pcDNA3 to replace wild type with mutant sequence. The mutations were verified by DNA sequencing. To generate KSHV BAC with LZ-deleted K-bZIP, we used a galK counterselection BAC system (). Briefly, we first replaced K-bZIP gene with a galK cassette (generated by PCR using the primers in ) based on BAC36 (). The recombinant bacteria (SW102) can only grow on minimal medium with galactose as the only carbon source and yield red colonies. Then the galK cassette was replaced by a PCR product that contains LZ-deleted or BAC-derived revertant K-bZIP DNA (generated by PCR using the primers shown in ). This is achieved by selecting against the galK cassette by resistance to 2-deoxygalactose on minimal plates with glycerol as the carbon source. 2-Deoxygalactose is harmless unless phosphorylated by functional galK. Phosphorylation by galK turns 2-deoxygalactose into 2-deoxygalactose 1-phosphate, a non-metabolizable and therefore toxic intermediate that killed non-transformants. The resultant KSHV BACs (BACdLZ and BACdLZRev) were further verified by DNA sequencing of K-bZIP gene. Luciferase Assay Cells were collected 24 h after co-transfection of K-bZIP- and/or HDAC2-expressing plasmids with a luciferase reporter gene directed by the promoter of ORF50 (genomic location: 8, Genbank TM accession number (), pORF50-luc), ORF59 (genomic location: 4, accession number, ), or OriLyt (genomic location: 2, accession number, pOriLyt-luc). PRL-TK was included in each transfection system as an internal control for the normalization of the DNA transfection. The Dual-Luciferase reporter assay system (Promega) was used to examine the responsiveness of the promoters to K-bZIP and/or HDAC2. Each assay was performed in triplicate, and the luciferase activity was normalized by the amounts of total protein. Transfection efficiency was normalized with the Renilla luciferase activities (pRL-TK). The cell lysates were assayed for firefly luciferase and Renilla luciferase activities by using a TD-20/20 luminometer with a dual autoinjector (Promega, Turner Designs). The luciferase assays were carried out according to the manufacturer's instructions (Promega). HDAC Activity Assay HDAC activity was assessed with the HDAC activity assay kit (Upstate-Millipore, Lake Placid, NY) according to the manufacturer's instructions. Immune complexes were incubated with 20,000 cpm [ 3H]acetyl-labeled histone H4 peptide (Upstate-Millipore, Lake Placid, NY) in 1× HDAC buffer at room temperature for 24 h with rolling. Reactions were stopped by adding 50 μl of 0.12 n acetic acid, 0.72 n HCl. The released acetate was extracted in 0.5 ml of ethyl acetate and mixed in 5 ml of scintillation solution, and radioactivity was measured in a scintillation counter. All assays were performed in triplicate. Antibodies Mouse anti-HDAC1 (ab31263), rabbit anti-HDAC1 (ab19845), rabbit anti-HDAC2 (ab32117), and mouse anti-HDAC2 antibodies (3F3, ab51832) were purchased from Abcam Inc. (Cambridge, MA; 1:250 for immunofluorescence; 1:1000 for Western blot). Monoclonal antibody against tubulin (T-9026) was purchased from Sigma-Aldrich (1:1000 for Western blot). Polyclonal antibody against PML (sc-5621), monoclonal antibody against Ac-histone H3 (Lys-24) (sc-34262), and monoclonal anti-enhanced GFP (sc-9966) were purchased from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA; 1:100 for immunofluorescence to show PML and 1:1000 for Western blot to probe enhanced GFP). Anti-Kb-ZIP (K8) was a gift from Dr. Yuan (University of Pennsylvania) (). Nick Translation Double-stranded DNA probes for in situ hybridization and Southern blot assays were labeled by nick translation as described previously (). Briefly, 1 μg of plasmid DNA (BAC36; the whole KSHV genome was cloned in the BAC vector, which was a gift from Dr. Gao) (), 10× nick translation buffer, 0.05 m m dNTP (dATP, dCTP, and dGTP), 0.01 m m dTTP, 0.04 m m biotinylated UTP, 1 unit of DNA polymerase I, and appropriate concentrations of DNase I were incubated at 15 °C for 50 min. Labeled fragments obtained from the protocol were 200–500 bases long as determined on 2% agarose gels. Immunocytochemistry and Fluorescence in Situ Hybridization For the immunofluorescence assay of adherent cells, cells were grown on round coverslips (Corning Glass Inc., Corning, NY) in 24-well plates (Falcon, BD Biosciences). For immunofluorescence assays in BCBL-1 cells, cells were washed with PBS, fixed with 1% paraformaldehyde, and cytospun to slides for the immunofluorescence assay, using different antibodies as desired. Cells were fixed in 1% paraformaldehyde (10 min at room temperature) and permeabilized in 0.2% Triton (20 min on ice) by sequential incubation with primary and Texas Red- or fluorescein isothiocyanate (FITC)-labeled secondary antibodies (Vector Laboratories, Burlingame, CA) for 30 min each (all solutions in PBS). For simultaneous detection of viral protein and specific DNA sequences, cells were first immunostained for viral proteins and then treated for 1 h at 37 °C with RNase (Roche Applied Science; 100 μg/ml in PBS) for the detection of DNA. After refixing in 4% paraformaldehyde (10 min at room temperature), samples were equilibrated in 2× SSC (1× SSC is 0.15 m NaCl plus 0.015 m sodium citrate), dehydrated in ethanol (70, 80, and 100% ethanol for 3 min each at 20 °C), air-dried, and incubated overnight at 37 °C with the hybridization mixture. For DNA detection, probe and cells were simultaneously heated at 94 °C for 4 min to denature DNA. After hybridization, samples were washed at 37 °C with 55% formamide in 2× SSC (twice for 15 min each), 2× SSC (10 min), and 0.25× SSC (twice for 5 min each). Hybridized probes were labeled with Texas Red-avidin (Vector Laboratories; 1:500 in 4× SSC plus 0.5% BSA), and signals were amplified by using biotinylated anti-avidin (Vector Laboratories; 1:250) followed by another round of Texas Red-avidin staining. Finally, cells were equilibrated in PBS, stained for DNA with Hoechst 33258 (0.5 μg/ml), and mounted in Fluoromount-G (Fisher Scientific). Immunoblot Analysis Proteins were separated by sodium dodecyl sulfate-7.5% polyacrylamide gel electrophoresis (SDS-PAGE) (10–20 μg loaded in each lane), transferred to nitrocellulose membranes (Amersham Biosciences), and blocked with 5% nonfat milk for 60 min at room temperature. Membranes were incubated overnight at 4 °C with primary antibody followed by an incubation with a horseradish peroxidase-coupled secondary antibody (Amersham Biosciences) and detection with enhanced chemiluminescence (Pierce). For the detection of protein in the immunoprecipitation, we used secondary antibodies from TrueBlot ULTRA (eBioscience; catalog number 18-8817 for mouse or 18-8816 for rabbit). Membranes were stripped with stripping buffer (100 m m β-mercaptoethanol, 2% SDS, 62.5 m m Tris-HCl, pH 6.8); washed with PBS, 0.1% Tween 20; and used to detect additional proteins. Preparation of Nuclear Extracts Nuclear extracts were obtained essentially as described previously (). Briefly, monolayer cells were washed with PBS and scraped into fresh Eppendorf tubes. Cell pellets were resuspended in cold buffer A (10 m m HEPES-KOH, pH 7.9 at 4 °C, 1.5 m m MgCl 2, 10 m m KCl, 0.5 m m dithiothreitol, 0.2 m m phenylmethylsulfonyl fluoride) and incubated at 4 °C for 10 min. After centrifugation, pellets were resuspended in cold buffer C (20 m m HEPES-KOH, pH 7.9, 25% glycerol, 420 m m NaCl, 1.5 m m MgCl 2, 0.2 m m EDTA, 0.5 m m dithiothreitol, 0.2 m m phenylmethylsulfonyl fluoride) by vortexing and incubated at 4 °C for 30 min. Clarified extracts were transferred to fresh tubes and stored at −70 °C until use. Coimmunoprecipitation Antibodies were coupled to protein G-Sepharose beads (Amersham Biosciences) according to the manufacturer's instructions. After a wash with PBS, 0.1% bovine serum albumin, beads were incubated overnight at 4 °C with clarified extracts; washed again in PBS, 0.1% bovine serum albumin; and resuspended in a mixture of PBS and 2× Laemmli buffer (20 μl of each). After heating at 95 °C for 5 min, beads were removed by centrifugation, and supernatants were analyzed by SDS-PAGE and immunoblotting. Viral DNA Isolation from Cells and Southern Blotting Viral DNA samples were isolated by Hirt's method with modification for KSHV genome-harboring cells (). The DNA was separated by running a 1% agarose gel and transferred onto a Nylon membrane (Bio-Rad). A biotin-labeled probe made from the full-length KSHV BAC DNA by nick translation was hybridized to the membrane. After the membrane was washed according to standard protocol, the hybridization signal was detected using an Ambion detection system (Ambion Inc., Austin, TX). Chromatin Immunoprecipitation (ChIP) Assay and Real Time PCR HEK 293T cells harboring BACdLZ, its BAC-derived revertant, or BAC36 were stimulated with TPA (20 ng/ml) and sodium butyrate (0.5 m m) for 24 h and fixed with 1% formaldehyde. A ChIP assay was performed using the EZChIP kit (Upstate-Millipore, Billerica, MA) according to the manufacturer's protocol. The amount of the DNA ChIPed by antibodies (normal IgG, anti-K-bZIP, and anti-HDAC2) was examined by real time PCR in a 25-μl reaction using the primers shown in that were designed according to Ref.. All samples were analyzed in triplicate using SYBR Green 2× Master Mix (Applied Biosystems, Foster City, CA) on an Applied Biosystems 7900HT Fast Real-Time PCR System. After an initial denaturation incubation of 5 min at 95 °C, 45 cycles of three-step cycling were performed with an annealing temperature of 60 °C. Melt curve analysis was then performed to verify product specificity. The relative ratio of Ct of each antibody-ChIPed DNA over respective IgG (rabbit IgG or mouse IgG)-ChIPed DNA was determined by examining the change in threshold cycle number (delta Ct) with standard deviation calculated to account for variance in both IgG- and antibody-ChIPed DNA signal. Confocal Microscopy Cells were examined with a Leica TCS SPII confocal laser-scanning system. Two or three channels were recorded simultaneously and/or sequentially and controlled for possible breakthrough between the green and red channels and between the blue and red channels. To balance the signal strength, we used Leica image enhancement software to scan the image to separate the signal from its background. Because of the variability among cells in any culture in terms of cellular size and shape, we selected the most typical cells that were photographed, and they are presented here at high magnification. HDAC1 and -2 Were Recruited into DNA Replication Domains with K-bZIP Cellular proteins begin interacting with viral proteins soon after infection. Previously, K-bZIP has been shown to colocalize with ND10 proteins, such as PML, which is known to defend against herpesviral infection (, ). We wished to explore whether the other cellular defensive proteins (especially HDAC family proteins) would interact with KSHV K-bZIP. First we examined the relationship of K-bZIP with HDAC1, HDAC2, and PML at early times after reactivation of BCBL-1 cells by TPA (). After 12 h of TPA treatment, the cells were stained with anti-K-bZIP and secondary antibody in red ( A, D, G, and J–L) and anti-HDAC1 ( B and J), anti-HDAC2 ( E and K), and anti-PML ( H and L) in green. We can see that upon reactivation by TPA KSHV latency (without treatment of TPA;, J, K, and L) was switched to the lytic stage, thus making K-bZIP detectable. K-bZIP formed small punctate dots ( A, D, and G). After merging ( C, F, and I), K-bZIP dots were shown to colocalize with ND10 (PML in green; G–I) but were not associated with HDAC1 ( A–C) or HDAC2 ( D–F) that diffuse in the nucleus. The colocalization of K-bZIP and PML was consistent with previous reports (, ). Distribution of K-bZIP and HDAC at early time of TPA treatment of BCBL-1. BCBL-1 cells were treated with TPA (20 ng/ml) for 12 h, then fixed, and permeabilized. Cells were stained first with primary antibodies to detect proteins as indicated and then with Texas Red-labeled secondary antibody. The primary antibodies include anti-K-bZIP ( A, D, and G), -HDAC1 ( B), -HDAC2 ( E), and -PML ( H). The panels show K-bZIP alone ( red; A, D, and G), HDAC1 alone ( green; B), HDAC2 alone ( green; E), PML alone ( green; H), and the merging of the two ( C, F, and I). Scale bar, 20 μm. The cells without TPA treatment ( J, K, and L) were also stained for both K-bZIP ( red) and HDAC1 ( J; green), HDAC2 ( K; green), or PML ( L; green). Previously, we also reported that K-bZIP is present in viral DNA replication domains (). Viral DNA replication happens at the late stage after the reactivation of BCBL-1 cells by TPA. As shown in, G–I, we performed DNA hybridization using a probe that was made from KSHV genomic DNA by nick translation. The BCBL-1 cells were stained with anti-K-bZIP antibody in red ( G), and the DNA is shown in green ( H). There are two cells in, G–I; the small green dots in the right cell represent the episomes (around 10 per cell on average) of KSHV DNA still in latency, which is similar to that in BCBL-1 cells not treated with TPA ( J). However, in the cell on the left, the green domain represents the viral DNA replication domain ( H). At this point, K-bZIP no longer exists as dots but instead as a large domain ( G) that overlaps the DNA replication domain ( I). The DNA replication of KSHV in BCBL-1 cells after treatment with TPA was also evidenced by Southern blot assay ( K), demonstrating that no variations in KSHV DNA signal can be detected without treatment with TPA, compared with increased KSHV DNA signal at 24 h after TPA treatment. Distribution of K-bZIP and HDAC at late time of TPA treatment of BCBL-1. A–J, BCBL-1 cells first were treated with TPA (20 ng/ml) for 48 h, then fixed and permeabilized, and finally stained with primary antibodies to detect proteins as indicated and then with Texas Red-labeled secondary antibody. The primary antibodies include anti-K-bZIP ( A, D, G, and J), -HDAC1 ( B), -HDAC2 ( E), and -PML ( H). The panels show K-bZIP alone ( red; A, D, and G), HDAC1 alone ( green; B), and HDAC2 alone ( green; E). KSHV DNA was shown by FISH ( green; H and J), and merging is shown in C, F, I, and J. Scale bar, 20 μm. K, Southern blot assay to examine the DNA replication in BCBL-1 cells after being treated or untreated with TPA. W/o, without. In all cells with observable DNA replication domains, K-bZIP formed domains that overlapped the DNA replication domains (). Therefore, K-bZIP domains can be used to show KSHV DNA replication domains. At 48 h after TPA treatments, BCBL-1 cells were fixed, and an immunofluorescence assay was performed to show HDAC1 and -2 () localization. HDAC1 and HDAC2 always form a complex together to deacetylate histones and modify DNA conformation into a tight structure, making it more difficult for transcriptional activators to contact their targets. We observed that both HDAC1 and -2 are recruited to K-bZIP domains (, C and F) some time between 12 and 48 h. These results suggest that cellular defensive proteins (HDAC1 and HDAC2) respond to the reactivation process of KSHV and might play a critical role in blocking lytic gene expression. K-bZIP Interacts with HDAC and Reduces HDAC Activity To determine the manner in which HDAC1 and HDAC2 are recruited into the DNA replication domains, we asked whether K-bZIP interacts with HDAC1 or HDAC2. Nuclear extracts were prepared from BCBL-1 cells with or without TPA treatment and incubated with protein G beads conjugated with anti-K-bZIP (mouse), anti-HDAC1 (mouse), or anti-HDAC2 (mouse) antibody or normal IgG from mouse as a control. Pulled down proteins were then detected by Western blot assays using anti-K-bZIP, -HADC1, and -HDAC2 antibodies. Because the molecular weights of HDAC1 and HDAC2 are close to that of the heavy chain of IgG, we used secondary antibodies from TrueBlot ULTRA to mask the band of IgG heavy chain. As can be seen in A, we discovered that K-bZIP interacted with HDAC1 and HDAC2 as they both can bind to the anti-K-bZIP-conjugated beads. Because K-bZIP was found to be related only to HDAC1/2 in DNA replication domains at the late stage () but not at the early stage (; debatable, not easily detected), it is also unknown whether the interaction of K-bZIP with HDACs was mediated by KSHV DNA replication. Next, we wondered what the consequence of the HDAC/K-bZIP interaction might be. To measure the HDAC2-specific deacetylation activity, cell lysates from BCBL-1 cells (treated or untreated with TPA) were immunoprecipitated with anti-HDAC2 antibody (because HDAC1 and -2 exist in the same complexes, we only used anti-HDAC2 antibody) or anti-K-bZIP antibody and used for the assay. The results showed that HDAC2-associated deacetylation activity decreased slightly after treatment with TPA for 48 h ( B). HDAC2 activity from the complex co-immunoprecipitated by anti-K-bZIP antibody was reduced significantly ( B). However, the K-bZIP-associated HDAC activity was reduced by an HDAC inhibitor, sodium butyrate, suggesting that the K-bZIP-bound HDAC(s) may retain at least partial deacetylation activity that is sensitive to sodium butyrate. A, interactions of K-bZIP with HDACs. Nuclear extracts were prepared from BCBL-1 cells (treated or untreated with TPA for 24 h). Mouse ( m) anti-K-bZIP, -HDAC1, and -HDAC2, and mouse IgG ( mIgG) were bound to protein G beads and incubated with nuclear extracts. The eluted protein complexes were detected by Western blot using antibodies to detect the respective proteins as indicated on the right. To avoid the heavy chain of IgG, we used secondary antibodies from TrueBlot ULTRA (eBioscience; catalog number 18-8817 for mouse or 18-8816 for rabbit). B, effect of K-bZIP on HDAC2 activity. BCBL-1 cells were untreated ( lanes 1 and 3) or treated ( lanes 2 and 4) with TPA for 48 h. A total of 1.2 mg of cell lysates was immunoprecipitated with anti-HDAC2 ( left) or anti-K-bZIP ( right) antibodies. One-third of the precipitant was assayed for deacetylase activity with or without 1 μ m sodium butyrate. The results shown for deacetylation activity are the averages of three independent assays. IP, immunoprecipitation. Of three assays was shown as the error bar. Leucine Zipper Domain, Not K-bZIP SUMOylation, Is Important for Interaction of K-bZIP and HDAC2 To determine whether KSHV DNA replication is required for the interaction of K-bZIP and HDAC2, we transfected a K-bZIP-expressing plasmid into HEK 293 cells free of the other components of KSHV. Nuclear extracts were prepared 24 h post-transfection and incubated with anti-K-bZIP antibody (mouse)- or anti-HDAC2 antibody (rabbit)-conjugated beads, and Western blot assays were performed to check proteins in the complexes ( A). As shown, HDAC2 can be pulled down by anti-K-bZIP antibody; K-bZIP can also be pulled down by anti-HDAC2 antibody. Therefore, K-bZIP interaction with HDAC2 was not dependent on KSHV DNA replication. Interactions of K-bZIP with HDAC2. A, nuclear extracts were prepared from HEK 293 cells that were transfected with wild-type (WT) pK-bZIP. Mouse ( m) anti-K-bZIP, mouse IgG, rabbit ( r) anti-HDAC2, and rabbit IgG were bound to protein G beads and incubated with nuclear extracts. The eluted protein complexes were detected by Western blot using antibodies to check the respective proteins as indicated on the right. B, HEK 293 cells were co-transfected with pgfpSUMO-1 and K-bZIP-expressing plasmids (mutation diagram is shown on the left) for 24 h. Cell lysates were assayed by Western blot using antibodies as indicated on the right. C, nuclear extracts were prepared from HEK 293 cells that were transfected with wild type or mutated pK-bZIP. Mouse anti-K-bZIP, mouse IgG ( mIgG), rabbit anti-HDAC2, and rabbit IgG ( rIgG) were bound to protein G beads and incubated with nuclear extracts. The eluted protein complexes were detected by Western blot using antibodies to detect the respective proteins as indicated on the right. D, immunofluorescence assay to show K-bZIP with deleted LZ domain distribution in the nucleus. IP, immunoprecipitation. SUMOylation can affect the location of a protein in the nucleus and its interaction with other molecules. K-bZIP can be SUMOylated, so we wondered whether SUMOylation affects the interaction of K-bZIP with HDAC2. The K-bZIP protein SUMOylation motif has been mapped to a single amino acid residue (lysine 158) (), and a SUMO interaction motif has been identified in amino acids 72–75 (). We constructed several deletion mutations and a point mutation of K-bZIP ( B), including pK8_K158R point mutant, pK8_dl72–75 (deleted SUMO interaction motif), pK8_dl122–132 (deleted nuclear localization signal), and pK8_dl205–219 (deleted leucine zipper domain). After co-transfection of these plasmids with pgfpSUMO-1 (SUMO-1 was N-terminally fused with GFP) into HEK 293 cells, the cell lysates were run in a Western blot assay using anti-GFP and anti-K-bZIP antibodies as indicated in B ( right). As can be seen, K-bZIPs with mutated SUMOylation target (K158R), deleted SUMO interaction motif, and deleted nuclear localization signal were not SUMOylated. However, K-bZIP with loss of SUMOylation can still be pulled down by anti-HADC2 antibody in a co-immunoprecipitation assay ( C, left). Therefore, SUMOylation of K-bZIP is not important for its interaction with HDAC1/2. To map out the domain that is essential for the interaction of K-bZIP with HDAC1/2, we deleted the leucine zipper domain, pK8_dl205–219. K-bZIP with a deleted leucine zipper domain can still be SUMOylated ( B, right) but loses the ability to interact with HDAC2 in the co-immunoprecipitation assay (as shown in C, right). Therefore, the leucine zipper domain is essential for the interaction of K-bZIP with HDAC2. We found that the distribution pattern of K-bZIP with deleted LZ also changed, losing the punctate nuclear pattern and becoming diffuse ( D). K-bZIP Protein Represses Several KSHV Lytic Promoters via Its Interaction with HDAC2 A comprehensive test of KSHV gene activation by ORF50 and K-bZIP proteins was conducted recently (). However, the effects of K-bZIP on important lytic stage promoters (including ORF50, ORF59, and OriLyt) were not clear. Previous unpublished experiments performed in our laboratory have repeatedly revealed that K-bZIP has inhibitory effects on the promoters of ORF50 and OriLyt, effects that were not evident in the global detection assays (). For that reason, we performed co-transfection of the luciferase reporter vectors using K-bZIP- or ORF50-expressing plasmids to test the effects of either K-bZIP or ORF50 on those promoters. Results showed that K-bZIP has repressive effects on the KSHV lytic promoters (ORF50 and OriLyt) but no significant effect on the ORF59 promoter ( A). Interaction of K-bZIP and HDAC2 with KSHV gene promoters and transcription regulation of OriLyt, ORF50, and ORF59 promoters by K-bZIP, ORF50, HDAC, and K-bZIP mutants. A, HEK 293 cells were co-transfected with reporter plasmids containing the firefly luciferase gene under the control of the OriLyt ( lower), ORF59 ( middle), or ORF50 ( upper) promoter with pcDNA3 (to normalize the input DNA amount), K-bZIP-, or ORF50-expressing plasmid. Renilla luciferase plasmid was included in each transfection as an internal control. At 24 h post-transfection, Dual-Luciferase assays were performed with the cell lysates of transfected cells. Relative luciferase activities were calculated by dividing the normalized firefly luciferase activity of each reporter by that of the pGL3 plasmid in pcDNA3-transfected cells. B, ChIP assay. HEK 293T cells harboring BAC36, BACdLZ, or its BAC-derived revertant were fixed with 1% paraformaldehyde at 24 h post-treatment with TPA (20 ng/ml) and sodium butyrate (0.5 m m). The whole cell lysates were sonicated to fragment the DNA. The DNA was then ChIPed using anti-HDAC2, anti-K-bZIP, or anti-Ac-histone H3 ( AcH3) antibody. The ChIP assay was performed using an EZChIP kit (Upstate-Millipore, Billerica, MA) according to the manufacturer's protocol. The ChIPed DNAs were finally examined by real time PCR using the primers amplifying promoters of ORF50, ORF59, and OriLyt (). The relative DNA quantities were calculated by comparing the PCR signals from normal IgG and specific antibodies with that from input. C, the same as in A but different plasmids (K-bZIP mutants) were applied for co-transfection. Results in A and C are mean ± S.D. From three independent assays. To know whether the K-bZIP or LZ-deleted K-bZIP could bind to the OriLyt or ORF50 promoters in HEK 293T cell lines, a ChIP assay using anti-HDAC2, anti-K-bZIP, or anti-acetylated histone (Ac-histone H3) antibody was performed. The ChIPed DNAs were then examined by real time PCR using the primers amplifying promoters of ORF50, ORF59, and OriLyt (). As shown in B, after comparing the PCR signals from normal IgG and specific antibodies with that from input, we found that both HDAC2 and K-bZIP bind to promoters of ORF50 and OriLyt as does Ac-histone H3 in HEK 293T cells harboring BAC36 and BACdLZRev ( B, upper and lower panels). Association of K-bZIP with the promoter of ORF59 was not obvious. Although the association of HDAC2 and Ac-histone H3 with all promoters is evident and was detected in HEK 293T cells harboring BACdLZ, the level of DNA ChIPed by anti-HDAC2 is significantly lower compared with that from cells with BAC36 and BACdLZRev ( B). That the effect is prominent only in ORF50 and OriLyt suggests that the K-bZIP leucine zipper domain must be important for K-bZIP to recruit HDAC2 to the promoters of ORF50 and OriLyt. Finally, we wanted to know whether the interaction of K-bZIP with HDAC2 is important for the repression of the two promoters by K-bZIP. We co-transfected the HEK 293 cells with the luciferase-tagged reporter plasmids with ORF50 or OriLyt with K-bZIP, non-SUMOylated pK-bZIPK158R, or LZ-impaired pK-bZIPdl205–219. As shown in C, pK-bZIP158KR represses ORF50 and OriLyt nearly as well as wild-type K-bZIP. However, pK-bZIPdl205–219 lost the ability to repress the two promoters. Given the result that the LZ domain is important for interaction with HDAC (), these results suggest that the repressive function of K-bZIP might be due to recruitment of HDAC2 to promoters of OriLyt and ORF50 for K-bZIP. Leucine Zipper Domain Is Necessary for K-bZIP to Interact with Promoters of ORF50 and OriLyt As reported above, K-bZIP affected KSHV promoters of ORF50 and OriLyt. We asked whether the effects of K-bZIP on the KSHV promoters require interaction with DNA and if so whether the HDAC could be brought to KSHV promoters by K-bZIP to play repressive roles on gene expressions. For that purpose, we constructed a KSHV BAC DNA with LZ-deleted K-bZIP gene and its BAC-derived revertant (BACdLZRev). The BAC DNAs were verified as follows. 1) XhoI digestion showed that the pattern of KSHV BACdLZ was indistinguishable from that of its BAC-derived revertant or BAC36 ( A). 2) PCR of K-bZIP gene showed a slightly smaller band from KSHVdLZ than that from its BAC-derived revertant or BAC36 ( B). 3) DNA sequencing showed that amino acids 205–219 were deleted in-frame (data not shown). KSHV BACdLZ and its BAC-derived revertant. A, a seamless (galK counterselection) BAC system was used to construct KSHVdLZ, which contains an LZ-deleted K-bZIP. XhoI digestion was followed by running an agarose gel to separate the DNA bands. The gel shows that BACdLZ has the same pattern as those of BAC36 and BACdLZRev. B, PCR using the primer to amply K-bZIP gene shows that the size from BACdLZ is slightly smaller that those from BAC36 and BACdLZRev. C, HEK 293T cells harboring BAC36, BACdLZ, and BACdLZRev were treated with TPA and sodium butyrate for 0, 12, 24, and 48 h, and the whole cell lysates were applied for Western blotting assay using antibodies against latency-associated nuclear antigen (LANA), RTA, K-bZIP, and ORF45. Tubulin was used for controlling the sample loading. D, co-immunoprecipitation assays were performed to determine the interaction of K-bZIP with HDAC2 using the nuclear extracts from HEK 293T cells harboring BAC36, BACdLZ, and BACdLZRev. Results show that K-bZIP with deleted LZ fails to interact with HDAC2 ( middle panel), whereas WT K-bZIP interacts with HDAC2 ( left and right panels). MIgG, mouse IgG; rIgG, rabbit IgG; IP, immunoprecipitation; r, rabbit; m, mouse. We then transfected the BAC DNA (BAC36, BACdLZ, or BACdLZRev) into HEK 293T cells to make HEK 293T cell lines harboring KSHV genomes: HEK 293T/BAC36, 293T/BACdLZ, and 293T/BACdLZRev. The transiently transfected cells were purified by cell sorting of a GFP marker so that nearly all cells were KSHV BAC-positive. After treatment with TPA for different times as indicated in C, we collected the whole cell lysate samples. KSHV proteins (K-bZIP, RTA, latency-associated nuclear antigen, and ORF45) were then analyzed by Western blot. As can be seen, HEK 293T cell lines harboring KSHV BACs can all express viral proteins that are important for viral replication, including K-bZIP. C shows that KSHV RTA expression is reduced during reactivation when LZ is deleted from K-bZIP in the BAC (the second row of Western blot), which could be caused by the loss of the repressive effect of K-bZIP on HDAC. Co-immunoprecipitation assays ( D) using anti-K-bZIP and anti-HDAC2 antibodies showed that LZ-deleted K-bZIP cannot interact with HDAC2; this is consistent with the results obtained in BCBL-1 cells and the transfection system ( and ). We wished to determine whether the LZ domain of K-bZIP and HDAC2 are important for KSHV to replicate in HEK 293T cells. Cells harboring BAC36, BACdLZ, or BACdLZRev were prepared in 6-well plates and treated with TPA for different times as indicated in ( upper panel). Cells were collected together with medium and treated with protease K, and total viral DNA was extracted. KSHV DNA from cells was quantified by real time PCR compared with an external BAC36 standard. As can be seen, KSHV-BACdLZ replicated at a lower level than its BAC-derived revertant and KSHV-BAC36, especially at the late time point of 48 h. This might suggest that the LZ is important for late (48 h) but not early (24 h or less) actions of K-bZIP. Taken together, the leucine zipper domain is important for K-bZIP to interact with some KSHV promoters and for KSHV replication in HEK 293T cells. The interaction of K-bZIP with HDAC has two different functions: 1) to repress HDAC activity as shown in B to favor viral replication and 2) to recruit HDAC to and repress promoters of ORF50 and OriLyt. However, the general effects of K-bZIP through interaction with HDAC are apparently positive for KSHV replication because abolishing interaction with HDAC by deleting the LZ domain produced a defective phenotype of viral gene expression and DNA replication. KSHV DNA replication in HEK 293T cells. Upper panel, HEK 293T/BAC36, /BACdLZ, and /BACdLZRev cells were treated with TPA and sodium butyrate for 0, 12, 24, and 48 h. Cells with medium were collected and treated with protease K, and the total viral DNA was extracted by phenol and chloroform and precipitated. The extracted viral DNA samples were used for real time PCR using primers to amplify ORF73 (). The DNA copies were determined by comparison with a standard KSHV BAC DNA (known concentration of the DNA). Lower panel, HEK 293T/BAC36, /BACdLZ, and /BACdLZRev cells were infected with lentivirus carrying shRNA against HDAC2 and selected with puromycin. The puromycin-resistant cells were assessed for HDAC2 production by Western blot assay as shown on the right, and tubulin was used to control the sample loading. The three types of cells were then stimulated with TPA for different times as indicated, and the viral DNA level was determined by real time PCR as in the upper panel. To further demonstrate that the effects of K-bZIP on KSHV DNA replication are connected to HDAC activity, we knocked down HDAC2 using small hairpin RNA carried by a lentivirus against HDAC2 (shRNA plasmid: sc-44262-SH) from HEK 293T cells harboring the three different BAC DNAs. The effect of the siRNA to specifically inhibit HDAC2 gene expression is shown by Western blot assay in, right, lower panel. Interestingly, the defectiveness of KSHVdLZ replication can be significantly rescued by the inhibition of HDAC2 gene expression as shown in, left, lower panel. Taken together, although the K-bZIP has a negative effect on some KSHV promoters, its general function in KSHV is enhancing viral DNA replication through inhibiting HDAC activity. DISCUSSION Interest in K-bZIP of KSHV originated from the fact that it is the positional homologue of Zta, which is a reactivator in EBV (,,,, ). Both Zta of EBV and K-bZIP of KSHV are related to the basic leucine zipper (bZIP) family of transcription factors; moreover, both genes are adjacent to another conserved transcription activator, RTA. However, after comparison of the functions and protein structures of K-bZIP of KSHV with Zta of EBV, it was recognized that the two proteins have very limited similarities (). First, KSHV K-bZIP alone cannot switch KSHV from latent to lytic infection (). Second, within the amino acid sequence, K-bZIP of KSHV and EBV Zta are not significantly homologous (). Finally, K-bZIP of KSHV lacks a basic DNA binding region adjacent to its dimerization domain, and it has not been demonstrated to interact with DNA directly (). Therefore, KSHV K-bZIP might have different functions than does EBV Zta. The role of K-bZIP in gene regulation has been widely investigated, and it is believed to be essential for KSHV reactivation. However, the part it plays in KSHV reactivation is not yet fully understood. Accumulated evidence shows that K-bZIP can interact with different cellular and viral proteins to present both trans-repressive and trans-activating activities (,,,, ). K-bZIP can interact with SUMO (), ND10 components (, ), CBP (, ), and CCAAT/enhancer-binding protein (). Although it has several functions, its primary function is to arrest cell cycle progression in the G 1 phase (, ); this is accomplished by its interaction with cellular proteins and results in the regulation of cell cycle protein production (, ). Its post-translational modifications (including SUMOylation) have also attracted tentative interest because they are related to the repressive effect of K-bZIP on gene regulation () and because in addition SUMOylation often aids protein interaction (). K-bZIP protein was discovered to inhibit TGF-β signaling through interaction with CBP (). CBP is an acetylase and modifies histone structure to loosen DNA conformation, making it more accessible for gene transcription factors. HDAC family proteins deacetylate histones and have the opposite function of CBP. Using a co-immunoprecipitation assay, we observed that K-bZIP can interact with HDAC1 and -2. And yet, both HDAC1 and HDAC2 are present in the DNA replication domain, and both colocalize with K-bZIP; for this reason, we believe that K-bZIP has additional, as yet unverified functions. The interactions of K-bZIP and HDAC were not dependent on KSHV DNA replication because K-bZIP interacts with HDAC in a co-transfection system. SUMO modification did not affect the interaction of K-bZIP with HDAC. Moreover, LZ-deleted K-bZIP failed to interact with HDAC, demonstrating that the leucine zipper domain is essential for the interaction. However, the functions of K-bZIP on KSHV promoters are not the same. On the one hand, in a co-transfection system using a luciferase assay, we found that the interaction of K-bZIP with HDAC is required for K-bZIP to perform its inhibitory function on KSHV promoters ORF50 and OriLyt. On the other hand, K-bZIP interaction with HDAC can directly repress the activity of HDAC, and a recent comprehensive study of the function of K-bZIP showed that it can activate 21 other KSHV promoters (). These data suggest that the repression by K-bZIP of some promoters may be important for the maintenance of latency, whereas later activation of other promoters will be part of the lytic reactivation pathway. In the established HEK 293T cells harboring KSHV BAC, after treatment with TPA, KSHV with a LZ-deleted K-bZIP had a reduced replication phenotype. This implies that the repressive effect of K-bZIP on HDAC activity is more important than its effect on some viral promoters, explaining why KSHVdLZ has a defective phenotype. Several herpesviral proteins have been discovered to interact with HDAC and to have different functions. CMV IE1 interacts with HDAC, represses deacetylase activity, and enhances viral replication (, ). However, IE2 of CMV interacts with HDAC2 and has repressive effects on several promoters (). Here, we are the first to report that KSHV K-bZIP interacts with HDAC, playing an important role in enhancing viral DNA replication. Its repressive effects on some important KSHV promoters (such as ORF50 and OriLyt) seem to contradict its importance in KSHV replication. We were curious whether the repressive effects of K-bZIP on promoters require interaction with HDAC because HDAC is generally an inhibitor of gene expression. ChIP assay results ( B) suggested that 1) HDAC2 can bind to the promoter of ORF59 that is not associated with K-bZIP, 2) the leucine zipper domain is important for the interaction of K-bZIP with the promoters of ORF50 and OriLyt that might be mediated by HDAC, and 3) the repressive effects of K-bZIP on KSHV promoters might work through a direct interaction with HDAC and promoters. The repressive effects of OriLyt and ORF50 on KSHV promoters did not result in lower KSHV replication (). The repressive effects of K-bZIP on HDAC activity are important to KSHV replication because the knockdown of HDAC2 partially complements KSHVdLZ replication in HEK 293T cells. In summary, we discovered that K-bZIP interacts with HDAC1/2; this interaction might be crucial for presenting HDAC1/2 in viral DNA replication domains. We found that the leucine zipper domain is essential for the interaction of K-bZIP and HDAC2 and that this interaction is independent of SUMOylation. We also provide evidence that K-bZIP is able to repress HDAC deacetylase activity and interact with and inhibit the lytic gene promoters (ORF50 and OriLyt) of KSHV. Our results suggest that K-bZIP might regulate KSHV gene expression through interacting with HDAC. Most importantly, our observations that the leucine zipper domain of K-bZIP is important for KSHV replication in HEK 293T cells and that K-bZIP can inhibit HDAC activity suggest that this inhibition of HDAC plays an important role in viral replication. Our model for the function of K-bZIP interaction with HDAC has two layers. 1) K-bZIP can bring HDAC molecules to some promoters, thereby having a repressive effect, and 2) it can bind to HDAC and inhibit the effects of the deacetylase, which in turn causes K-bZIP to have a positive effect on KSHV replication. Footnotes • * This work was supported, in whole or in part, by National Institutes of Health Grant U54RR022762 from the National Center for Research Resources (to Q. This work was also supported by RCMI Program Pilot Grant 2G12RR003050-24 (to Q. T.), American Cancer Society Grant RSG-090289-01-MPC (to Q. T), and American Cancer Society Institutional Research Grant IRG-92-032-13, Subaward 60-14599-01-01-S6 (to Q. • 2 The abbreviations used are: KSHV Kaposi sarcoma-associated herpesvirus HDAC histone deacetylase SUMO small ubiquitin-like modifier LZ leucine zipper BAC bacterial artificial chromosome RTA replication and transcription activator CBP cAMP-response element-binding protein (CREB)-binding protein PML promyelocytic leukemia protein TPA 12- O-tetradecanoylphorbol-13-acetate ChIPed chromatin immunoprecipitated EBV Epstein-Barr virus bZIP basic leucine zipper. • Received October 21, 2011. • Revision received March 12, 2012. • © 2012 by The American Society for Biochemistry and Molecular Biology, Inc. → → → Installing a Compatible Theme Installing a Compatible Theme Archived file. Good only up to BP 1.5 version After you activate BuddyPress, you’ll get this message under the header of the Plugins page: BuddyPress is ready. You'll need to to take advantage of all of the features. We've bundled a default theme, but you can always. Activate a BuddyPress compatible theme This link goes to your admin themes page where you can activate the bp-default theme to test drive the different BuddyPress components that you, as Super/Site Admin, chose to enable for your community. Note: If you want to modify or customize the bp-default theme, instead to ensure that your revisions will not be overwritten when you upgrade BuddyPress. Install some other compatible themes This link goes to a special theme installation page listing all BuddyPress-compatible themes and child themes of the bp-default theme which have been accepted in the WordPress Themes repository and are ready to install and activate in your site. Return: The Theme Manual. BuddyPress themes are generally only used on the blog that is running BuddyPress plugin (this is normally the main blog on a WordPress MU site). Your new BuddyPress theme is activated as follows: 1. Login to your dashboard. Go to Appearance > Themes. Click on Activate below your. Home Blog WordPress Plugins 10 BuddyPress Plugins You Must. There are tons of great options you can activate. Since it was designed to pick up your theme. No themes found. Try a different search. About; Blog; Hosting; Donate; Support; Developers; Get Involved; Learn; Showcase; Plugins. 
BuddyPress themes and bp-default child themes are also available at: • • 2. Make sure that the theme you want to install is compatible with the WordPress/BuddyPress versions you have activated in your installation. • BP 1.1.3 compatible themes are not compatible with BP 1.2+ installations. • New functions/features contained in WP 3.0 compatible themes will not work with WP/MU 2.9.2 installations Upgrade your existing WordPress theme This link goes to a new BuddyPress admin panel where you can automatically install the BP Template Pack plugin to integrate BuddyPress component templates into your active WordPress theme. If you’re an avid WordPress user, then you may have heard of the well known social networking solution called. BuddyPress is a great way to build a social network for your company, school, sports team, or any niche community. 
This article is meant to be used as a guide to get you started off in the right direction with BuddyPress and WordPress. In this Introduction to BuddyPress, I will cover how to install the software, the useful features and interface BuddyPress supplies, BuddyPress extensions and much more. A Closer Look BuddyPress is a social network that allows users sign-up and start creating their very own profiles.  
Users can send messages, make connections with other users, create groups or communities, share status updates and much more. BuddyPress, as defined by its creators, is a “ social network in a box “. If your company, community, school, or anything of that nature needs a place to interact with each other then look to BuddyPress as an outstanding solution. BuddyPress comes with several components that integrate directly with your existing WordPress site. The great advantage to BuddyPress over other solutions is the fact that you don’t need to have a customized theme to use it. BuddyPress creates a few new pages and options within your theme and WordPress admin area. Other than these additions, your site can function just the same as it always has. Benefits To Using BuddyPress • Complete social networking solution in one plugin. • Themeable and adaptable to almost any WordPress theme. • Extendable. Forums and additional plugins can all be added and maintained very easily. • User friendly. All the features of your favorite sites like Facebook and Twitter are yours to do with as you please. • Users can register and contribute thus creating a larger audience for your website. • A community can be established which builds up hype about your website. Installation Installing BuddyPress is easy. You actually install it like any other plugin. Simply head to your WordPress admin area and click on Plugins. Search for BuddyPress. The first result should be the installation you are looking for. You will notice a lot of plugins during your search. There are a ton of add-ons that make BuddyPress even more useful. I will cover a few shortly. BuddyPress successfully installed. After a full installation of the BuddyPress plugin, you will likely notice a few changes to the way your admin area looks. In the top right, where you username and gravatar lives, you will notice a new circle with a zero inside. This is a notifications indicator. With BuddyPress you can opt to have settings to include notifications for messages, comments, friend requests and more. If you hover over your username and gravatar, you will notice a much more elaborate drop down menu of available options. User Dropdown and new links that appear after installing BuddyPress. Here you see what my current installation and profile drop down looks like. Notice the new options for activity, profile, notifications, messages, friends, groups, and settings. You can also edit your profile and update your account information just like on sites such as Facebook or Twitter. You should also notice many of the features you probably use today on social networking sites are now right within your website. Finally if you look to the left side navigation you will notice two new links to Activity and Groups (depending on your settings). Users BuddyPress revolves around its user base. Without users, your BuddyPress installation might be a little lacking. The first step, once you have enough users, is to allow your new users to register for their own account. In order to allow people to register, you first need to make sure you actually allow this process within your main WordPress settings panel. By default WordPress is set to disallow user registration. You can even assign new registrars different roles within your WordPress site. For BuddyPress, you will likely want to set your default user role to Subscriber or Contributor settings for maximum security. Set your membership settings to “Anyone can register” to allow BuddyPress to work properly. Find this settings in the General Settings panel. Once registered, each user is given their own profile and admin area to control. They can modify their account information as well as keep track of their time on the site. The login to their account will be the familiar WordPress login screen you already use to access your admin area. The big difference is that once the new registrars login, the settings and actions they can control will be limited based on the role you have assigned. Adding a new user can be done by the user themselves or by clicking the register button once directed to do so. The screen below displays the Login and Register links in the bar atop your website. With a bit CSS, you can style these links to fit in more with the look of your website. List of current user base For this walk-through I am using our new. For the sake of brevity I haven’t made a lot of customizations to the theme as you can probably tell. There is so much more you can customize when using this theme, but I won’t be covering that today. For our purposes, I will just be exploring how well BuddyPress adapts with the theme as well as the functionality of the plugin itself. Profile If you’ve used WordPress before and are familiar with the various user roles, then editing your profile should be both an easy and familiar process. After logging in you can click your user drop down menu at the top right side of your screen. Click on Edit Profile and you will be taken to a page where you can update things such as your nickname, admin color style, biographical information, as well as include a link to your website. You can also update your password if needed. Below is the current setup on my testing WordPress Installation. Remember that some of this information gets displayed to other users who register on your site. Having a memorable username and gravatar thumbnail helps other users recognize you and identify with you, so be sure and choose good credentials. User Profile with a single status update. Within this page, which every user has, are a large number of features including: • Last Activity • Profile • Notifications • Messages • Friends • Groups • Settings It’s safe to say that this page is a dashboard of sorts for BuddyPress. Users can message each other, post status updates, join groups, update notification settings as well modify their global settings. Requirements To use BuddyPress successfully and to its full potential, the plugin requires a few pages to be present on your website. If you look in your WordPress Dashboard under Settings > BuddyPress you will see a Pages tab similar to the one shown below. BuddyPress Pages Settings Area. By default the Activity, Groups, and Members pages are create. You will need to create both the Register and Activate pages if you want to allow users to register on your site. If you are using BuddyPress, then this is a requirement. Once all the pages are present and you have allowed anyone to register to your site within your main settings panel, you should be ready to use BuddyPress the way it was intended. Widgets By default, BuddyPress includes some custom widgets that can offer really useful information to all of your sites users. These a used for logging in, notifications, and also allow you to seeing who is online and logged in at the same time as you. The default widgets provided by BuddyPress. BuddyPress Toolbar Plugin Homepage The BuddyPress Toolbar is almost a must have if you decide to implement BuddyPress on your website. The toolbar is a an extension to BuddyPress which gives admins quick access to the latest activity, custom settings, and even more to the websites owner or moderator. Think of it a short cut plugin that is a huge time saver if your site happens to grow and have a lot of users to maintain. If you search for BuddyPress when adding a new plugin you’ll see the BuddyPress Toolbar coming in at second on the list. BbPress plugin homepage bbPress is a software package built with forums and WordPress in mind. The plugin seamlessly adds a great forums solution to WordPress’s already powerful content management system. This saves you time and headache while searching for a third party solution of a similar caliber. The seamless integration I described above also happens to work with BuddyPress. Alongside WordPress, you can download both BuddyPress and bbPress and have an amazingly cool website in just a few clicks. To install it, navigate to the new plugin page and search for bbPress. Install the plugin and click activate. Head to your admin area and then to the Settings / Forums. You should see a screen similar to the one below. At the very bottom of this screen you should see a section titled BuddyPress Integration. The title speaks for itself here. This section controls the integration between your users who signed up with BuddyPress and the newly installed bbPress plugin. BuddyPress Links. Plugin that easily converts rich media for you when linking to it within BuddyPress. BuddyPress Links adds rich media sharing features to BuddyPress. If you have ever shared a link on Facebook or GooglePlus, you may have noticed the link automatically displays some of the data you are actually linking to. Data such as images, video, thumbnails, and excerpts are what typically gets displayed. The BuddyPress Links plugin make this task simple and easy. Simply download and activate the plugin and you and your users should be good to go. There is a free version and a pro version. The pro version gives you access to group integration, link re-sharing, Dailymotion and Vimeo support and more. BuddyPress Docs. Write documentation with user collaboration with BuddyPress. BuddyPress Docs is a plugin geared towards a collaborated documentation environment. Think of it as wikipedia but on a smaller scale. The ability to have other users contribute is also a nice add on. Conclusion WordPress alone is a very powerful platform. Combining WordPress with BuddyPress makes the platform even more powerful. Within minutes your WordPress website can transform into a complete social network where users discuss, relate, comment, and improve whatever it is your service or product(s) provide. Using BuddyPress can allow you to open new doors and target a much larger audience. With a few customizations and a couple plugins properly activated and updated, there’s no telling how much you can achieve. I hope you’ve learned a bit about BuddyPress and how it can be useful on some websites. If your site or company needs a place to communicate, then look no further than BuddyPress + WordPress. Great article, I have used Buddypress a few times and really like it but bbpress really lets it down in a few areas. The biggest of which is it’s lack of ‘View Latest Posts’ functionality. Trying to get people to move from systems like SMF and phpbb is very difficult when they can’t see all new posts since their last visit with a single click. Very frustrating and has resulted in us dropping it (and therefore Buddypress) in favour of a forum system. This then brings other headaches like bridging users between wordpress and the forum chosen but at least the users will use it!! Cheers Gavin •. Thanks for a great intro to Buddypress/bbpressan hour ago a client bowled up demanding bb-style forums, and I was sat tryijng to decide between bbpress and Buddypress. Now I’m well on my way, although I should just say, there don’t seem to be any good get-you-started guides out there, even amongst the documentation released by buddypress/bbpress themselves. Everyone seems to assume installers are fully up-to-date with FB/etc so know what to expect – not me 8(. Any good links? Like a walk-through of how a user would interact with these services? Thanks again either way, great post. This has been a major bummer for me. I bought Divi thinking it could really be the last theme I ever buy. But I make a LOT of BuddyPress sites too, and Divi just plain sucks with BuddyPress. I keep seeing Elegant Themes say that BP is a 3rd-party plugin and therefore isn’t supported. Technically it’s not a part of WP core, sure, but it’s OWNED by Automattic!! It’s the #4 featured Plugin in the depository! BuddyPress, along with bbPress, are the two most basic plugins that EVERY WP theme that exists should support!!! If Divi simply worked well with BP, it would make so many people insanely happy. Better yet, if Divi gave some basic styling to BP pages, or was able to give some modules that worked with BP (so people could make more custom profiles and group pages), sales for DIVI would easily double overnight. It’s remarkable to me that they just don’t seem to care about BP at all 🙠x81 •. 
To get achievements in Half-Life 2 with cheats (ie: sv_cheats set to '1'), you activate sv_cheats and whatever cheats you want (god, impulse 101, noclip, etc.), save your game, exit out of Half-Life 2, relaunch Half-Life 2, and reload your save. Sv_cheats 1 only disables achievements for the session you activated cheats. Ending that session re-enables achievements. Your save game still has the cheats activated, but because the new game session doesn't realize that, achievements are still enabled. This page contains a list of cheats, codes, Easter eggs, tips, and other secrets for Half-Life 2: Black Box for PC. If you've discovered a cheat you'd like to add to the page, or have a correction, please. (Portal) Aperture Science Login You can find a neat Easter egg hidden in Test Chamber 17. One malfuncitoning wall panel reveals a den with cameras and pictures of cubes. Right of the piston and near the floor of is a user-name and password. Once you have written that down go to www.aperturelabrat oies.com. And type login. Enter the user-name and password. Type 'Help' if you want to know all you can do. 
ApertureScience.com The various grafitti in Portal can help you crack the code for the website www.aperturesc ience.com. Go to the website (it uses Shockwave Flash) and type in the command LOGIN. The user name is CJOHNSON and the password is TIER3. Once you are in, you may type in HELP for a list of valid commands. Addition by PRO_GAMER_: You should also be able to login to aperturescience.com with the username GLADOS and the password PORTAL, as well. Half-Life Cheat Codes - PC. If this is the first time you are enabling cheats they will not activate until you load a game. /developer 2; Half-Life. The best place to get cheats, codes, cheat codes, walkthrough, guide, FAQ, unlockables, tricks, and secrets for Half-Life 2: Episode Two for PC. Noclip: Toggle. Half Life 2 Cheat Codes includes God mode and all of the known cheats for the shooter and provide that extra edge needed to win the game. Noclip Allows you to fly trough your level without player collisions. Requires sv_cheats to be set to 1. Developer Developer mode enables more verbose console. Addition by red7ring: It actually doesn't matter what name you login with. You can login with any name you want. It's the password you insert that's important. Addition by Spilled Salt: After logging in, type at the prompt 'thecakeisalie' (minus the quotes). Doing so will unlock some secret messages for you to view. Half-life 2 Portal Gun Hack This process modifies your game files, so make back- ups of the proper files and directories before attempting this hack. • You need both Portal and Half-life 2 for starters. Install or re-install both games now. • Back up your Portal directory. Unless you changed the install path, the directory is in.Steamsteamapps(your_username)portal. If you make a mistake during this hack, you may need to re-download Portal through Steam (or at worse, Half- life 2 through Steam). • Download and install GCFScape if you do not have it already. This program allows you to access the cached game files for games using the Source engine. • Head into the directory.Steamsteamapps for all the available GCF archives available. Open the file half-life 2 content.gcf via GCFScape. Click the hl2 folder on the left pane of the GCFScape Explorer window. Right mouse click maps and select the command Extract to place all the contents of the maps folder into the directory.Steamsteamappsthe_great_bundiniportalportal. This merges Half-life 2 maps with the Portal map folder. • Repeat the above step with the scenes folder via GCFScape. • Boot up Steam to run Portal. Drop the developer console with ‘~’ (you may need to enable it under your keyboard options or enable it via the -console command switch on the executable's shortcut). • On the developer console, type maps • to list all the available maps; this should inclde the Half-life 2 maps imported from the previous steps. Type map and then the name of that map (i.e., map d1_canals_01). • Once the map is loaded and you can play, drop the console and enable the cheats for Portal by entering sv_cheats 1. Give yourself all (including the Portal Gun) by entering impulse 101. You may need to input upgrade_portalgun to enable you to fire both portals. • Enjoy the havoc. Note that this hack also works with and Episode 2. You will notice that while NPCs from Half-life cannot pass through portals, all other inert objects (including yourself) and fire will pass through with no problems. Submitted by Primotechnology.com Half-Life 2 Cheats Turn on the cheat console in-game, and press the tilde (~) to bring it up. You can then type the codes in below for the desired effect. • ai_disable - NPCs are turned off • give item_battery - Receive 15 battery points • give item_healthkit - Receive 25 health points • kill - Character eliminates himself • npc_create - Create NPCs Hit tilde “~” and type 'npc_create npc_ [name]” to spawn an NPC. Here is a list of the names you can enter for [name]: • 'alyx' (Alyx Vance) • 'barney' (Barney Calhoun) • 'dog' (Dog) • 'kleiner' (Dr. • Adobe livemotion 1.0: LVW1-292 • Adobe photoshop 6.0: PWW6-948 • Adobe golive 5.0: GJW5-460 • Adobe illustrator 9.0: ABW91-999-830 • Adobe premiere 6.0: MBW6-881 • Adobe photoshop 6.0: PWW6-948 • Adobe Photoshop 8 – serial: EWW4-030-259 • Ad-aware Professional – password: 1291453 • Advanced Office XP Password Rec. Pro 2.0 AOXPPR-PRO-TADFH-43369-HTHAF-42745 • Advanced Paradox Password Recovery 1.01 MSPP-83852-DWMRA-762 • Advanced PDF Password Recovery 1.34 PDFP-99322-EYSVY-886 • AlamDV Name: KHS Inc.| s/n: dqonbaobazyxwvut • AlamDV 2.01 name: Claudio Duffini| s/n: bspdopxmaziansxm • Alcohol 120% v1.3.1.823 s/n: • AL12PPPPAFAE4rneMMz. • Harry Potter And The Chamber Of Secrets! Kumpulan Serial Number Koleksi Serial Number (SN,S/N) Software/Serial Number Collection Win XP Home OEM: JQ4T4-8VM63-6WFBK-KTT29-V8966 Win. WINDOWS XP – Media Center Edition 2004*GERMAN* SN: KFX2B-HXQ68-78RWH-RPXPK-68DHM 14 460. Partition Magic 8.0 SN: PM800FR1. MAGIX Photo Manager 15 Free Download Activation Code. Media player, and. A confirmation with your login data together with activation code to unlock MAGIX.    
|
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
